 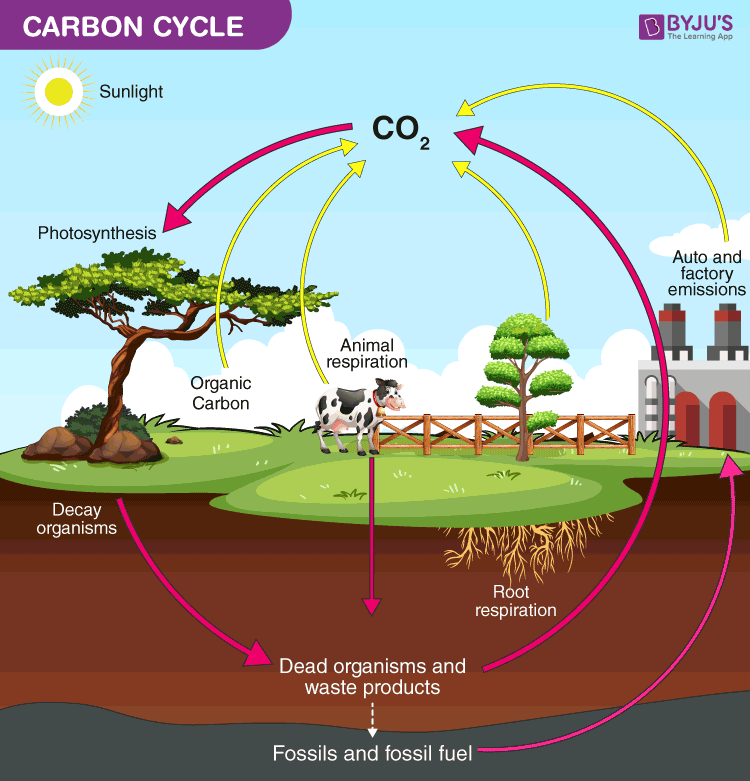 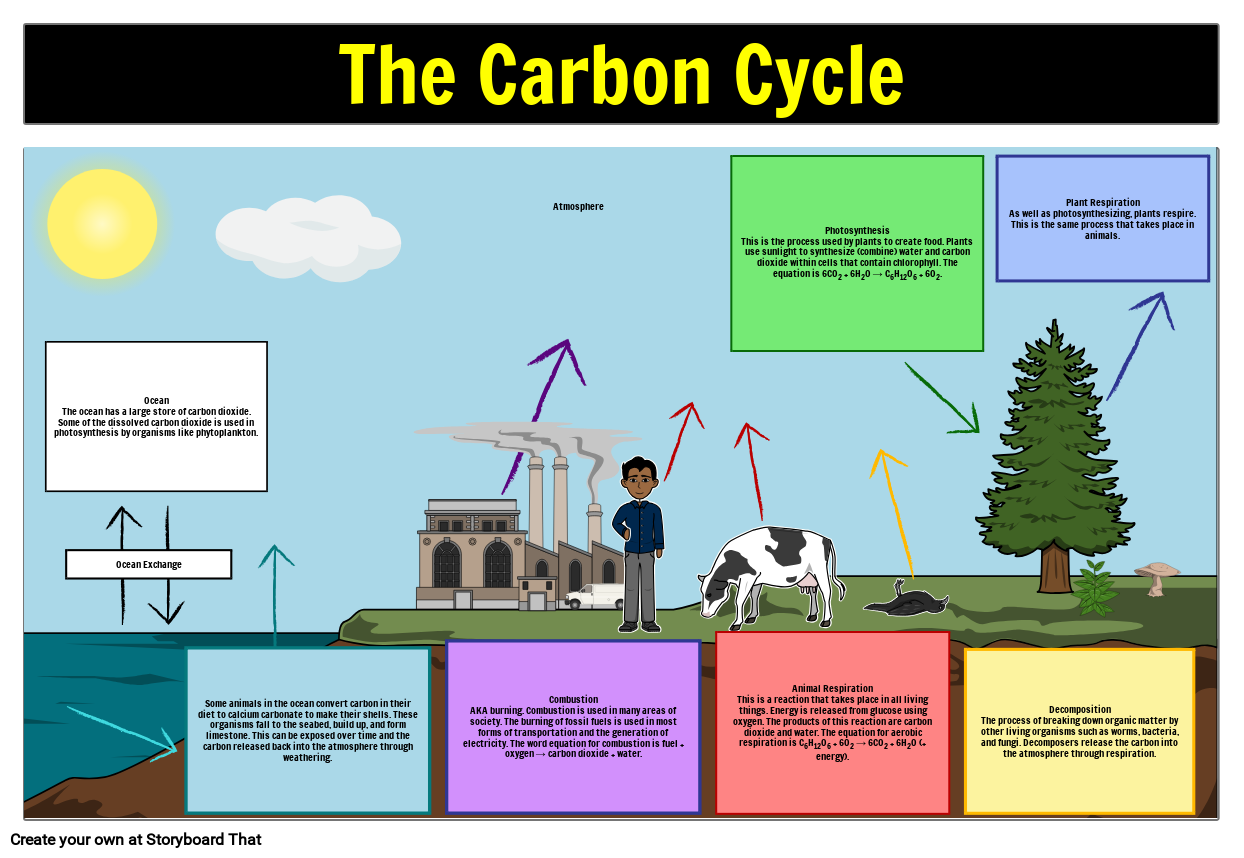
Applying this same logic to carbon tetrachloride, CCl 4, yields oxidation numbers of +4 for carbon and −1 for chlorine. And so, the oxidations numbers for oxygen and hydrogen in water are −2 and +1, respectively. Imagine that the polarization of shared electrons within the O−H bonds of water were 100% complete-the result would be transfer of electrons from H to O, and water would be an ionic compound comprised of O 2− anions and H + cations. The sum of the partial negative and partial positive charges for each water molecule is zero, and the water molecule is neutral. Consequently, H atoms in a water molecule exhibit partial positive charges compared to H atoms in elemental hydrogen. The shared electrons comprising an O−H bond are more strongly attracted to the more electronegative O atom, and so it acquires a partial negative charge in the water molecule (relative to an O atom in elemental oxygen). Water is a covalent compound whose molecules consist of two H atoms bonded separately to a central O atom via polar covalent O−H bonds. Calcium fluoride, CaF 2, is comprised of Ca 2+ cations and F − anions, and so oxidation numbers for calcium and fluorine are, +2 and −1, respectively.Ĭovalent compounds require a more challenging use of the formalism. Sodium chloride, NaCl, is comprised of Na + cations and Cl − anions, and so oxidation numbers for sodium and chlorine are, +1 and −1, respectively. Simple ionic compounds present the simplest examples to illustrate this formalism, since by definition the elements’ oxidation numbers are numerically equivalent to ionic charges. To illustrate this formalism, examples from the two compound classes, ionic and covalent, will be considered. Consequential to these rules, the sum of oxidation numbers for all atoms in a molecule is equal to the charge on the molecule. For an atom in a compound, the oxidation number is equal to the charge the atom would have in the compound if the compound were ionic. By this description, the oxidation number of an atom in an element is equal to zero. The oxidation number of an element in a compound is essentially an assessment of how the electronic environment of its atoms is different in comparison to atoms of the pure element. Oxidation Numbersīy definition, a redox reaction is one that entails changes in oxidation number (or oxidation state) for one or more of the elements involved. Readers wishing additional review are referred to the text chapter on reaction stoichiometry. Since reactions involving electron transfer are essential to the topic of electrochemistry, a brief review of redox chemistry is provided here that summarizes and extends the content of an earlier text chapter (see chapter on reaction stoichiometry). Balance chemical equations for redox reactions using the half-reaction method.Identify the oxidant and reductant of a redox reaction.Describe defining traits of redox chemistry.By the end of this section, you will be able to: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
